Monitoring and Management > 4. The Atmosphere >
Describe ozone as a molecule able to act both as an upper atmosphere UV radiation shield and a lower atmosphere pollutant
- Ozone is found at very low levels in the lower atmosphere (0.02 ppm in clean air).
- The formation of ozone through the interaction between sunlight, hydrocarbons and nitrogen oxides can lead to higher concentrations in the lower atmosphere or troposphere.
- Ozone is poisonous to humans (as well as most other organisms), with its effects in larger than normal concentrations including:
- Breathing problems.
- Aggravation of respiratory problems.
- Headaches.
- Fatigue.
- Death (in high concentrations).
Structure of ozone
- Ozone exists at much higher levels in the upper atmosphere or stratosphere (ranging from 2 ppm to 8 ppm in the stratosphere), where 90% of the atmosphere’s oxygen is found.
- In the upper atmosphere both oxygen gas and ozone gas absorb different parts of the ultraviolet radiation range in a cycle of ozone formation and decomposition:
-
- Oxygen absorbs high energy ultraviolet radiation as part of the ozone formation process:
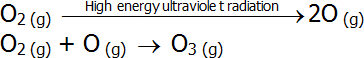
-
- Ozone absorbs medium energy ultraviolet radiation as part of the ozone decomposition process:
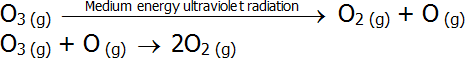
-
- In both of these sets of reactions, the second reaction is exothermic, which accounts for the increasing temperature in the stratosphere.
- Thus, the ozone layer acts as a radiation shield in the upper atmosphere by absorbing medium and high energy ultraviolet radiation.
Ozone-induced discolouration of a leaf




