The Acidic Environment > 5. Esterification >
Identify the IUPAC nomenclature for describing the esters produced by reactions of straight-chained alkanoic acids from C1 to C8 and straight-chained primary alkanols from C1 to C8
- Every ester has a unique two-word name.
- When an ester is produced from an alkanol and an alkanoic acid, the nomenclature is as follows:
- The alkanol reactant forms the first part of the name, with its suffix changing from “anol” to “yl”.
- The alkanoic acid forms the second part of the name, with the word “acid” being omitted, and its suffix changing from “oic” to “oate”.
- For example:
- The ester produced from methanol and ethanoic acid is known as methyl ethanoate.
- The ester produced from octanol and hexanoic acid is known as octyl hexanoate.
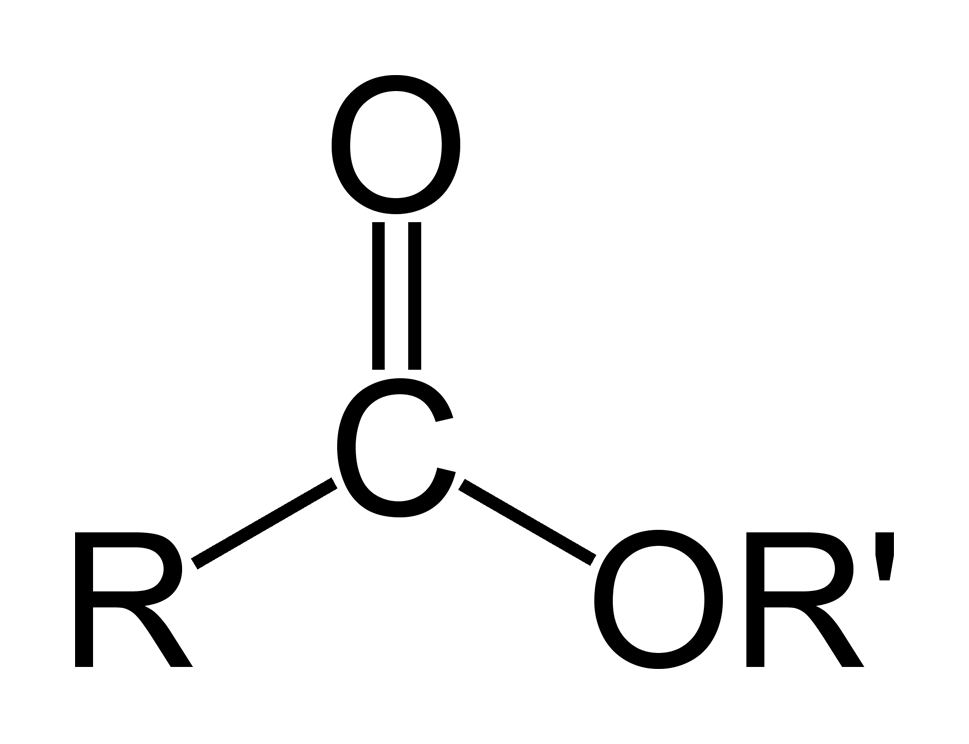
General formula of an ester