The Acidic Environment > 5. Esterification >
Describe the purpose of using acid in esterification for catalysis
- Esterification: The process in which an alcohol and an acid are converted to an ester and water.
- Esterification is an example of a condensation reaction.
- Esterification reactions are equilibrium systems:
- Most esterification reactions involve a carboxylic acid.
- As alkanols are alcohols and alkanoic acids are acids, the reaction between an alkanol and an alkanoic acid is an example of esterification.
- For example, when ethanol and ethanoic acid are heated with a concentrated sulfuric acid catalyst, ethyl ethanoate and water are formed:
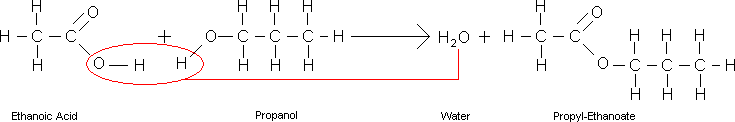
- For example, when propanol and ethanoic acid react, propyl ethanoate and water are formed: